Environmental concerns
Authors: Sean Cabaniss, [2014] David Park, [2014] Maxim Slivinsky, [2014] Julianne Wagoner, [2014] and Michelle Spiezio [2015]
Steward: David Chen, Jian Gong, and Fengqi You
Date Presented: Feb. 23, 2014
Overview
All industrial processes produce waste in some form that must be carefully handled and disposed of according to regulations set down by governments at varying levels. The key areas for consideration are emissions to the land, air, and water, waste management, smells, noise, and visual impact. It is important to remember that pollution considerations are both a moral and legal obligation for any engineer or industry [1]. Emissions from chemical plants are regulated by both local and federal governments. Plants are required to monitor and document waste streams discharged to the environment, maintain permits from local governmental agencies, and either pay fines or directly address any violations [2].
Human Health, Social and Economic costs
There are several costs associated with pollution from chemical plants. Governmental regulations are in place in order to minimize the human costs of waste streams. These costs come in the form of endangerment or direct harm to local communities. The most obvious cases include poisoning drinking water or making air difficult to breathe. The United States government has tried to keep these factors under control, but the governments of other countries often try not to pass similar legislations in order to attract more business. In these cases, engineers can be forced to make an ethical decision on whether to minimize waste even if not required to by law.
Social costs are often linked to human ones and involve the public perception of the company. A recent example of high social costs is the outcry in the wake of the BP oil spill. Companies caught or known for high pollution levels often receive bad press, which can also give investors a bad impression and hurt stock prices.
The final cost involved with waste considerations is the economic costs. In the short run, it may often be more profitable to do as little as possible to mitigate waste. However, in the long run it can often be in a company's best interest to take these factors into consideration because an optimized process or a process that makes use of recycle streams effectively will produce less waste while also saving money for the company. In addition, recovery facilities that separate waste streams are less expensive than waste treatment equipment [4].
Types of Pollution and Treatment
Noise Pollution
Causes & Effects
Treatment
Thermal Pollution
Causes & Effects
Treatment
Gas
Causes & Effects
Gas pollutants often come from fired heaters, boilers or flares in the form of CO2 or other sulfates and nitrates, particulates (PM10, PM2.5), Volatile Organic Compounds (VOC) and, less commonly, chlorine compounds [1]. These gaseous wastes are often under careful scrutiny from governments as well as the public since gases being released from towers are often the easiest forms of pollution for a casual observer to see.
Gas emissions can be particularly dangerous because left unchecked they can have far reaching consequences. Historically, the main focal points for gas emissions have been the effects on acid rain, ozone and greenhouse gases. Lately, greenhouse gases have been the most scrutinized due to growing concerns of global warming.
Treatment
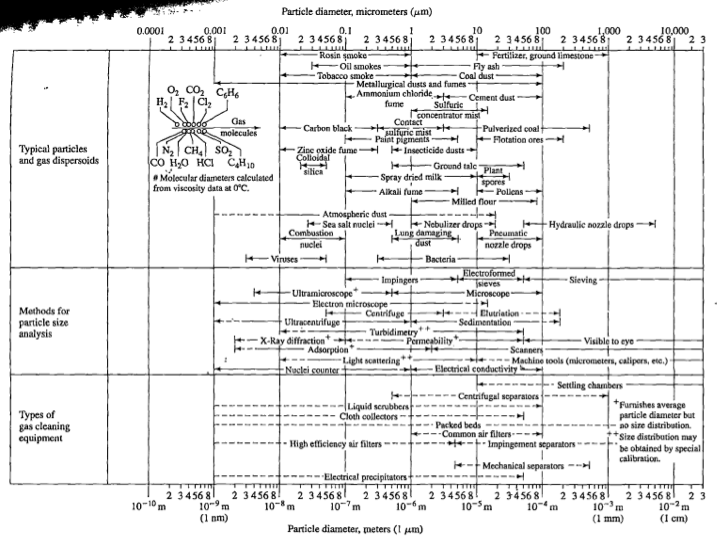
Gaseous wastes must be treated before being released into the atmosphere, especially given the recent emphasis on green processes and the growing alarm with regards to global warming. Due to the properties of gases, air pollution can be the most expensive and energy intensive. Noxious elements can often be removed by absorption or adsorption, which are the most common ways to treat gaseous wastes. Typically, there are two main classifications of air pollution control equipment: those that can physically remove particles and those associated with removing gaseous pollutants. Figure 3 compares the size and type of common particles to the method of particle analysis and appropriate cleaning equipment [4].
Figure 2:
Liquid
Causes & Effects
Water pollution typically comes from three streams: process waste, utility waste and run-off streams. Process waste streams are the unwanted side products of reactions and include salt water, hydrocarbon contaminated water, biologically contaminated water, and water with too high or low pH. Utility waste is mainly produced from cooling tower water blowdown. The purpose of blowdown is to prevent a buildup of salts in the towers, but produces a stream of contaminated water. Finally, run-off streams come from rain water, hydrant flushing and equipment washing.
Liquid wastes are typically not as far reaching as gaseous waste, but can be much more devastating. Ecosystems can be destroyed from lax wastewater policies. Local populations can suffer from a lack of clean drinking water or dying wildlife in the area. The BP oil spill was a highly visible incident that showed how much damage can be done by water borne pollutants.
Treatment
The main factors that usually have some sort of legislative constraints are pH levels, suspended solids, toxicity, and biological oxygen demand. Oxygen demand is often under looked as a form of pollution because there is rarely visible evidence that something is wrong in the stream. However, many local governments mandate that the water leaving a plant must have enough oxygen to sustain aquatic life before being released.
Liquid wastes are usually flammable and can be burned in an incinerator as long as care is taken to insure the temperatures are high enough to destroy the harmful compounds. This only shifts the problem, however, as it creates gaseous wastes.
Aqueous wastes can be the most immediately harmful to the environment and people living nearby and must be sent immediately to an effluent treatment. Some of the most common, harmful water contaminants are ammonia, salts from deionizers, hydrocarbons, spent acids, caustics, and various forms of biological contamination. Utilities on-site can also create large wastewater flows that must be purged in order to prevent solid buildups. The most efficient way to deal with contaminated runoff is to collect and purify it on-site before disposing of it [3].
Solid
Causes & Effects
Solid, hazardous wastes are generally the easiest to control, but can also be the most difficult to safely discard; once created they can last much longer and do more damage than liquid or gaseous pollution. Table 1 below shows various types of solid wastes and how they can be treated. A more detailed description of solid waste treatment can be found in the Treatment section below.
Table 1:
Solid wastes are generally identified as flammable, toxic, corrosive or reactive. Waste containing any of these characteristics must be specially handled to ensure as little damage to the local environment as possible. Left unchecked, large amounts of flammable waste could ignite and become very difficult to control. Toxic, corrosive or reactive materials have averse affects to any local populations and must also be disposed of with special care.
Treatment
Figure 1 shown above introduces several forms of solid waste disposal. The four main types are incineration, landfills, land treatment and recycle. Incineration can be one of the more expensive, but if done properly can potentially recover some fuel value. Landfills are useful for disposing large quantities of waste, but if the contents are legally classified as "hazardous", the waste must go through a pretreatment process in order to reduce the contaminants to an acceptable level. Finally, recycle can be the most profitable solution instead of disposal if the waste can be reused in another capacity.
Waste Minimization
Chemical engineers need to keep several environmental considerations in mind when designing processes. During the design of the process, the toxicities of all the products, byproducts, and wastes produced by the system must be considered. Then, the reaction pathway that minimizes toxic components should be chosen whenever possible. A good design will seek to minimize, recycle, and make use of segregated waste streams in order to satisfy the demands of the waste market [3]. Optimization is a powerful tool for waste minimization because it cuts down on toxic materials as well as improving the overall system. Optimization can be used to account for environmental impact, such as the effects of carbon monoxide or other greenhouse gases. The general 5-step process for waste minimization can be summarized as: 1) Identify waste components for regulatory impact. 2) Identify waste streams for size and economic impact. 3) List the root causes of the waste streams. 4) List and analyze modifications to address the root causes. 5) Prioritize and implement the best solutions [1].
Legislative Action
1972 Federal Water Pollution Control Act
The US Water Pollution Control Act set forth three major new regulations in an effort to keep toxic contaminants from getting into water supplies. Its goal was to achieve clean water for swimming, boating, and protecting fish and wildlife by 1983. This legislation said the EPA would set water quality standards for pollutants in surface water, set effluent guidelines for each industrial sector, and that it is unlawful to discharge any pollutant into navigable waters without a permit. [5]
Resource Conservation and Recovery Act (RCRA)
The ultimate goal of this act is to protect the groundwater from contamination. The act addressed waste management from "cradle to grave". It identified wastes on regulatory lists or if it has a certain characteristic level of flammability, toxicity, corrosivity, or reactivity. The act also specified how hazardous wastes must be labeled and tracked in transport as well as the treatments required for low levels of contaminants. [6]
Example Environmental Design Problem
Volatile Organic Compound Abatement [4]
The 1990 Clean Air Act mandates a reduction in emissions of volatile organic compounds (VOC). Any VOC emission sources exceeding 10tons/year must retrofit abatement processes with the best available control technology (BACT).
A paint spraying plant emits VOCs from vents in the paint spray booths. The stream contains primarily toluene, methyl ethyl ketone (MEK), and xylene, with small impurities of silicone and phosphorus. The concentration of VOCs in the dryer effluent varies between a minimum of 0.3 wt% VOC and a maximum of 1.2 wt% VOC with an approximate composition of 50% toluene, 25% MEK, and 25% xylene.
The painting company has commissioned you to evaluate three different technologies for a reduction in VOCs: thermal incineration, catalytic incineration, and carbon absorption and destruction of the VOCs. The low quality steam can be used by a nearby bottle washing plant.
Design an emission control plant for 50,000 scfm of vent gas at 100F and 25% relative humidity for 99% removal. The plant is located in Dearborn, Michigan, and the paint spray booths operate on a single 12-hour shift per day. Include the necessary start-up controls. The available fuel is natural gas or oil. Calculate the capital and operating cost and the $/lb or ton of VOC removed. Compare the three processes and recommend which is most suitable for this application.
Conclusion
Nearly all industrial chemical processes have pollution in one form or another. There are several sources and compounds of waste in all phases to be aware of and to know how to dispose of properly. A proper understanding of these hazardous compounds and materials as well as the effects these pollutants can have is essential, especially as an engineer. A chemical engineer's duty is to ensure that the waste, whether a gas, liquid, or sold is disposed of or recycled in a responsible manner and according to the local and federal guidelines. Proper waste management not only reflects well on a company and its engineers, but also can have lasting benefits to profits and the wellbeing of locals in the area. On the other hand, reckless waste handling policies are not only dangerous for a plant and those around it, but are in danger of serious repercussions from the government and in the EPA. Finally, engineers have a moral obligation to uphold certain standards even when working in countries with less strict waste management standards.
References
- Towler, G.P. and Sinnot, R. (2012). Chemical Engineering Design: Principles, Practice and Economics of Plant and Process Design. Elsevier.
- Towler, G.P. (2012). Chemical Engineering Design, PowerPoint presentation.
- Seider, W.D. (2004). Process Design Principles: Synthesis, Analysis, and Evaluation, Wiley: New York.
- E. Robert Becker, "Volatile Organic Compound Abatement", Environex, January 1994
- U.S. Government page "Digest of Federal Resource Laws of Interest to the U.S. Fish and Wildlife Service" Digest of Federal Resource Laws of Interest to the U.S. Fish and Wildlife Service
- EPA Website "Resource Conservation and Recovery Act (RCRA)" http://www.epa.gov/agriculture/lrca.html